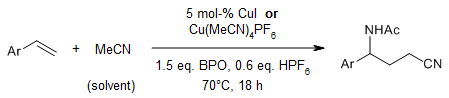Benzoyl Peroxide (BPO)
Product Manager:Nick Wilde

Benzoyl peroxide is a chemical compound with structural formula (C6H5−C(=O)O−)2, often abbreviated as (BzO)2. In terms of its structure, the molecule can be described as two benzoyl (C6H5−C(=O)−, Bz) groups connected by a peroxide (−O−O−). It is a white granular solid with a faint odour of benzaldehyde, poorly soluble in water but soluble in acetone, ethanol, and many other organic solvents. Benzoyl peroxide is an oxidizer, which is principally used as in the production of polymers.

Recent Literature
A direct asymmetric benzoyloxylation of aldehydes with benzoyl peroxide catalyzed by (S)-2-(triphenylmethyl)pyrrolidine provides optically active α-benzoyloxyaldehydes as useful chiral building blocks.
T. Kano, H. Mii, K. Maruoka, J. Am. Chem. Soc., 2009, 131, 3450-3451. https://doi.org/10.1021/ja809963s

The direct asymmetric α-benzoyloxylation of β-ketocarbonyls catalyzed by a chiral primary amine demonstrates excellent enantioselectivity for a broad range of substrates, which allows convenient access to highly enantioenriched α-hydroxy-β-ketocarbonyls.
D. Wang, C. Xu, L. Zhang, S. Luo, Org. Lett., 2015, 17, 576-579. https://doi.org/10.1021/ol503592n
A highly regio- and enantioselective hydroxyamination of aldehydes with in situ generated nitrosocarbonyl compounds from hydroxamic acid derivatives was realized by combining TEMPO and BPO as oxidants in the presence of a binaphthyl-modified amine catalyst.
T. Kano, F. Shirozu, K. Maruoka, J. Am. Chem. Soc., 2013, 135, 17735-17738.
https://doi.org/10.1021/ja4099627
Copper-catalyzed carboamination of alkenes enables an efficient synthesis of γ-amino butyric acid (GABA) derivatives from alkenes. In this difunctionalization reaction, acetonitrile serves as the source of the carbon and nitrogen functionalities. A copper-catalyzed radical-polar crossover mechanism is proposed.
N. Zhu, T. Wang, L. Ge, Y. Li, X. Zhang, H. Bao, Org. Lett., 2017, 19, 4718-4721. https://doi.org/10.1021/acs.orglett.7b01969
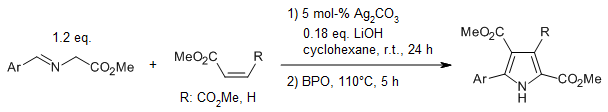
A one-pot sequential 1,3-dipolar cycloaddition/aromatization reaction sequence enables the synthesis of multisubstituted pyrroles from simple, easy available alkenes. A well-defined silver-catalyzed 1,3-dipolar cycloaddition reaction is followed by a benzoyl peroxide-mediated oxidative dehydrogenative aromatization reaction.
Y. Liu, H. Hi, X. Wang, S. Zhi, Y. Kan, C. Wang, J. Org. Chem., 2017, 82, 4194-4202. https://doi.org/10.1021/acs.joc.7b00180
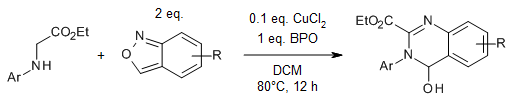
A convenient copper-catalyzed oxidative cross-dehydrogenative [4 + 2]-cyclization of glycine derivatives with anthranils enables an efficient and atom-economical synthesis of various 3,4-dihydroquinazolines. The reaction offers high efficiency and wide substrate tolerance.
J. Ren, C. Pi, Y. Wu, X. Cui, Org. Lett., 2019, 21, 4067-4071. https://doi.org/10.1021/acs.orglett.9b01246
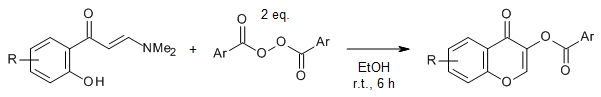
The use of aroyl peroxides as coupling partners enables a direct acyloxylation of the α-C(sp2)-H bond in tertiary β-enaminones under catalyst-free conditions and ambient temperature via a thermoinduced free-radical pathway. The present method offers an efficient synthesis of both acyloxylated chromones and enaminones.
Y. Guo, Y. Xiang, L. Wei, J.-P. Wan, Org. Lett., 2018, 20, 3971-3974. https://doi.org/10.1021/acs.orglett.8b01536
Photocatalysis enables the construction of 3-acyl-4-arylcoumarins in good yields from simple aldehydes and ynoates in the presence of inexpensive 2-tBu-anthraquinone as catalyst. The reaction proceeds through generation of an acyl radical intermediate, that reacts with ynoate, and then cyclization.
K. Kawaai, T. Yamaguchi, E. Yamaguchi, S. Endo, N. Tada, A. Ikari, A. Itoh, J. Org. Chem., 2018, 83, 1988-1996. https://doi.org/10.1021/acs.joc.7b02933
Quoted from:
https://en.wikipedia.org/wiki/Benzoyl_peroxide
https://www.organic-chemistry.org/chemicals/oxidations/benzoylperoxide.shtm
Aladdin:https://www.aladdinsci.com