Diethylsilane
Sandra Forbes
Product Manager

Recent Literature

Reduction of secondary amides to imines and secondary amines has been achieved using low catalyst loadings of readily available iridium catalysts such as [Ir(COE)2Cl]2 with diethylsilane as reductant. The stepwise reduction to secondary amine proceeds through an imine intermediate that can be isolated when only 2 equiv of silane is used. This system shows high efficiency and an appreciable level of functional group tolerance.
C. Cheng, M. Brookhart, J. Am. Chem. Soc., 2012, 134, 11304-11307.
DOI: 10.1021/ja304547s
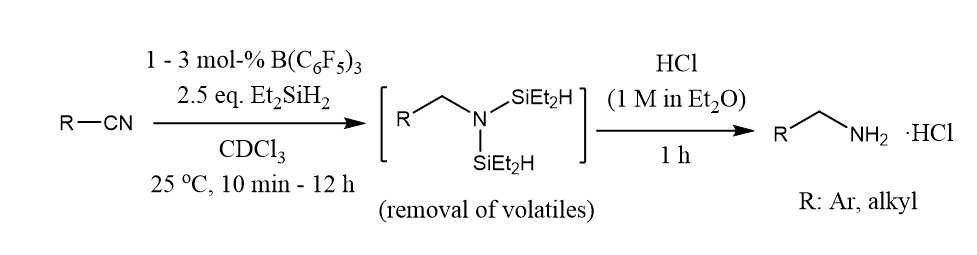
Silylative reduction of nitriles under transition metal-free conditions converts alkyl and (hetero)aryl nitriles efficiently to primary amines under mild conditions. The use of sterically bulky silanes enabled a partial reduction leading to N-silylimines.
N. Gandhamsetty, J. Jeong, Y. Park, S. Park, S. Chang, J. Org. Chem., 2015, 80, 7281-7287.

In an operationally convenient, mechanistically unique protocol, Lewis base activation of silyl acetals generates putative pentacoordinate silicate acetals, which fragment into aldehydes, silanes, and alkoxides in situ. Subsequent deprotonative metalation of phosphonate esters followed by HWE with aldehydes furnishes enoates.
U. S. Dakarapu, A. Bokka, P. Asgari, G. Trog, Y. Hua, H. H. Nguyen, N. Rahman, J. Jeon, Org. Lett., 2015, 17, 5792-5795.
DOI: 10.1021/acs.orglett.5b02901
After an iridium-catalyzed reduction of secondary amides, the resulting imines can undergo the Strecker reaction, the Mannich reaction, allylation, and [3+2]-cycloaddition. The method shows high chemoselectivity in the presence of other functional groups such as methyl ester.
Y. Takahashi, R. Yoshii, T. Sato, N. Chida, Org. Lett., 2018, 20, 5705-5708.
DOI: 10.1021/acs.orglett.8b02421
B(C6F5) enables a metal-free hydrogenative reduction of substituted N-heteroaromatics using hydrosilanes as reducing agents. The optimized conditions were successfully applied to quinolines, quinoxalines, and quinoline N-oxides. The initial step in the catalytic cycle involves 1,4-addition of the hydrosilane to the quinoline to give a 1,4-dihydroquinoline followed by (transfer) hydrogenation to deliver the tetrahydroquinoline.
N. Gandhamsetty, S. Park, S. Chang, Synlett, 2017, 28, 2396-2400.
Quoted from: https://www.organic-chemistry.org/chemicals/reductions/diethylsilane.shtm
Aladdinsci: https://www.aladdinsci.com/


