Tert-Butyl peroxybenzoate, TBPB

Product Manager:Nick Wilde

Recent Literature
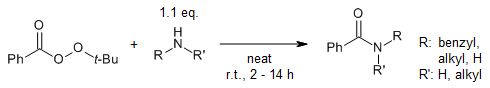
The reaction of tert-butyl peroxybenzoate (TBPB) and ammonia/amines provides the corresponding primary, secondary, and tertiary amides under catalyst- and solvent-free conditions in excellent yields. TBPB is an efficient and highly chemoselective benzoylating reagent for aliphatic amines in the presence of aromatic amines and hydroxyl groups.
D. K. T. Yadav, B. M. Bhanage, Synlett, 2015, 26, 1862-1866.
https://doi.org/10.1055/s-0034-1380811

A mild protocol for the α-C-H cyanation of tertiary aliphatic, benzylic, and aniline-type substrates and complex substrates tolerates a broad range of fuctional groups, including various heterocycles and ketones, amides, olefins, and alkynes. The presented catalyst system especially tolerates functional groups that typically react with free radicals, suggesting an alternative reaction pathway.
O. Yilmaz, C. Dengiz, M. H. Emmert, J. Org. Chem., 2021, 86, 2489-2498.
https://doi.org/10.1021/acs.joc.0c02642
A radical-promoted decarboxylation enables a transition-metal-free phosphorylation of cinnamic acids with P(O)H compounds under mild conditions. This method provides simple, efficient, and versatile access to a broad range of valuable (E)-alkenylphosphine oxides in good yields.
L. Liu, D. Zhou, J. Dong, Y. Zhou, S.-F. Yin, L.-B. Han, J. Org. Chem., 2018, 83, 4190-4196.
https://doi.org/10.1021/acs.joc.8b00187
A convenient and general atom transfer radical addition (ATRA) of simple nitriles, ketones, and esters to alkynes provides a wide range of β,γ-unsaturated nitriles, ketones, and esters.
Y. Xiao, Z.-Q. Liu, Org. Lett., 2019, 21, 8810-8813.
https://doi.org/10.1021/acs.orglett.9b03444
A convenient and general atom transfer radical addition (ATRA) of simple nitriles, ketones, and esters to alkynes provides a wide range of β,γ-unsaturated nitriles, ketones, and esters.
Y. Xiao, Z.-Q. Liu, Org. Lett., 2019, 21, 8810-8813.
https://doi.org/10.1021/acs.orglett.9b03444
Visible-light irradiation mediates the synthesis of alkynylsilanes in very good yields from alkynes and silanes in the presence of CuCl as precatalyst. A large scale flow reaction and late-stage functionalization of natural products show the potential of the transformation.
Q.-C. Gan, Z.-Q. Song, C.-H. Tung, L.-Z. Wu, Org. Lett., 2022, 24, 5192-5196.
https://doi.org/10.1021/acs.orglett.2c02022
An efficient I2/TBPB mediated oxidative formal [4+1] cycloaddition of N-tosylhydrazones with anilines represents a simple, general, and efficient approach for the construction of 1,2,3-triazoles under metal-free and azide-free conditions.
Z.-J. Cai, X.-M. Lu, Y. Zi, C. Yang, L.-J. Shen, J. Li, S.-Y. Wang, S.-J. Ji, Org. Lett., 2014, 16, 5108-5111.
https://doi.org/10.1021/ol502431b
An oxidative cascade cyclization strategy enables an unprecedented construction of thiazole-2-thiones from enaminones via a cascade of C(sp2)-H/C(sp2)-H bond sulfurations and C(sp3)-H bond thiocarbonylation. This transformation allows for the efficient synthesis of thiazole-2-thiones with broad tolerance in moderate to excellent yields from simple enaminones with elemental sulfur.
B. Zhan, D. Liu, Y. Sun, Y. Zhang, J. Feng, F. Yu, Org. Lett., 2021, 23, 3076-3082.
https://doi.org/10.1021/acs.orglett.1c00751
A nickel-catalyzed reaction of acrylamides or enamides with diazoacetates provides indolin-2-ones or 1,4-dicarbonyl compounds in the presence of a peroxide and ethanol as hydrogen source and solvent.
J. Zhao, P. Li, Y. Xu, Y. Shi, F. Li, Org. Lett., 2019, 21, 9386-9390.
https://doi.org/10.1021/acs.orglett.9b03610
Quoted
from:https://www.organic-chemistry.org/chemicals/oxidations/tert-butyl-peroxybenzoate-tbpb.shtm
Aladdin:https://www.aladdinsci.com







